How FMN and FAD Precisely Regulate Protein Metabolism in Dairy Cows?
Part II: The Central Role of Riboflavin in Rumen Microbial Protein Synthesis and the Methionine Cycle
Protein metabolism is one of the most critical physiological processes determining growth performance, health status, and milk protein yield in dairy cows. At the molecular level, this process is tightly regulated by riboflavin-derived coenzymes—flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD). These flavins function as indispensable redox regulators, coordinating microbial protein synthesis in the rumen and amino acid metabolism in the host animal.
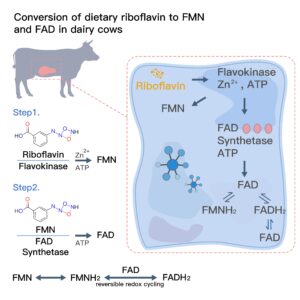
1. Conversion of Riboflavin into FMN and FAD in Dairy Cows
Riboflavin exerts its biological activity only after being converted into its coenzyme forms. This conversion involves two ATP-dependent enzymatic steps:
- Flavokinase catalyzes the phosphorylation of riboflavin to form FMN
- FAD synthetase subsequently adenylates FMN to produce FAD
The efficiency of these reactions is influenced by the cow’s dietary energy supply and trace mineral status. For example, zinc acts as an activator of flavokinase, linking riboflavin metabolism to overall mineral nutrition.
A defining feature of FMN and FAD is their reversible redox capability. The oxidized forms (FMN, FAD) accept hydrogen atoms and electrons to become FMNH₂ and FADH₂, and then return to their oxidized states after electron transfer. This cycling enables continuous participation in metabolic reactions without being consumed, making flavins ideal regulators of protein-related enzymatic pathways.
2. Precise Regulation of FMN and FAD on Protein Metabolism in Cattle
2.1 Importance of Microbial Protein for Dairy Cows
Rumen microorganisms convert dietary nitrogen and non-protein nitrogen (NPN), such as urea, into microbial protein. This microbial protein can contribute 30–50% of the total absorbable crude protein available to dairy cows, making it a major determinant of protein utilization efficiency.
However, microbial protein synthesis is highly sensitive to coenzyme availability. FMN and FAD function as prosthetic groups for numerous microbial enzymes, including:
- Amino acid synthases
- Transaminases
- Oxidoreductases involved in nitrogen assimilation
2.2 Role of FMN and FAD in Microbial Enzyme Activity
Flavin-dependent enzymes are directly involved in the synthesis of amino acids such as alanine and glutamate, which serve as precursors for peptide and protein formation within microbial cells. Adequate riboflavin supply enhances the synthesis of FMN and FAD, thereby:
- Increasing the activity of flavin-dependent enzymes
- Promoting microbial growth and protein synthesis
- Improving the conversion efficiency of dietary nitrogen
3. FAD and the Methionine Cycle
3.1 Methionine as a Limiting Amino Acid for Milk Protein Synthesis
Beyond the rumen, protein metabolism in dairy cows is further regulated at the level of host amino acid metabolism. Among essential amino acids, methionine is often the first limiting factor for milk protein synthesis. Insufficient methionine supply has been associated with an 8–12% reduction in milk protein yield in high-producing dairy cows.
3.2 FAD-Dependent Regulation of One-Carbon Metabolism
FAD plays a pivotal role as a coenzyme for methylenetetrahydrofolate reductase (MTHFR), a key enzyme in the methionine cycle. MTHFR catalyzes the conversion of:
- 5,10-methylenetetrahydrofolate → 5-methyltetrahydrofolate
This reaction is the central step in one-carbon metabolism, ensuring the stable transfer of methyl groups. The resulting 5-methyltetrahydrofolate serves as a methyl donor for the remethylation of homocysteine to methionine.
4. Synergistic Effects of Riboflavin and Folic Acid
Riboflavin and folic acid exhibit a well-documented nutritional synergy in dairy cows:
- Folic acid functions as the carrier of one-carbon units
- FAD, derived from riboflavin, ensures the activation and recycling of folate coenzymes
When both nutrients are adequately supplied, the efficiency of the methionine cycle can increase , resulting in:
- Improved amino acid balance
- Enhanced milk protein synthesis
- Better metabolic resilience in early lactation
This synergy is particularly important for high-yielding dairy cows, whose demand for methyl groups and amino acids is substantially elevated.
Conclusion
FMN and FAD, the biologically active derivatives of riboflavin, play a decisive role in regulating protein metabolism in dairy cows. Through their dual action in rumen microbial protein synthesis and the host methionine cycle, these flavin coenzymes enhance nitrogen utilization efficiency, support milk protein synthesis, and improve overall production performance.
References
Historical Discovery and Early Research Literature (1879-1935)
1. Blyth, A. W. (1879). On the yellow pigment of milk whey (lactochrome). Journal of the Chemical Society, Transactions, 35, 532-535. https://doi.org/10.1039/CT8793500530
2. Chick, H., & Roscoe, M. H. (1928). The dual nature of water-soluble vitamin B. II. The effect upon young rats of vitamin B2 deficiency and a method for the biological assay of vitamin B2. Biochemical Journal, 22, 790-799. https://doi.org/10.1042/bj0220790
3. Kuhn, R., & Wendt, H. (1933). Über Lactoflavin, ein Vitamin B2-haltiges Pigment aus Milch. Berichte der Deutschen Chemischen Gesellschaft, 66, 1262-1267. https://doi.org/10.1002/cber.19330660823
4. Karrer, P., et al. (1933). Über die Isolierung von Lactoflavin in reiner Form aus Milch. Helvetica Chimica Acta, 16, 1059-1064. https://doi.org/10.1002/hlca.19330160327
5. Kuhn, R., et al. (1933). Über Ovoflavin, ein Vitamin B2-haltiges Pigment aus Eiern. Berichte der Deutschen Chemischen Gesellschaft, 66, 1301-1308. https://doi.org/10.1002/cber.19330660828
6. Kuhn, R., et al. (1935). Über die Struktur des Lactoflavins (Riboflavin). Berichte der Deutschen Chemischen Gesellschaft, 68, 2067-2074. https://doi.org/10.1002/cber.19350681226
7. Kuhn, R., et al. (1935). Über die Synthese des Riboflavins (Vitamin B2). Angewandte Chemie, 48, 177-182. https://doi.org/10.1002/ange.19350480402
8. McCormick, D. B., & Greene, T. J. (2012). The discovery and characterization of riboflavin. Annals of Nutrition & Metabolism, 61, 224-230. https://doi.org/10.1159/000343111



